BridgeBio keeps FDA approval spree rolling, this time with nod for targeted bile duct cancer med | Fierce Pharma

Neil Kumar - founder and CEO of BridgeBio, superforecaster in rare disease drug development - YouTube

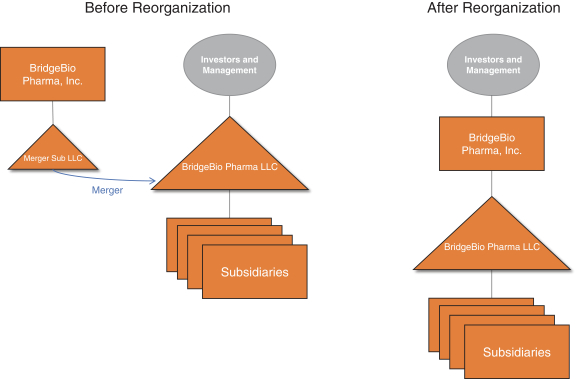
BridgeBio Pharma Licenses Late-Stage Oncology Drug Infigratinib to Tackle FGFR-Driven Maladies; Establishes New Subsidiary QED Therapeutics with $65 Million in Initial Financing

BridgeBio Pharma on Twitter: "We are thrilled to have received our first international approval for our first cancer therapy – paving the way for patients in Canada to receive our medicine targeting